What to Know Before You Invest in Psychedelics Companies Stock in 2023

A Primer on Psychedelic Medicine
After many decades of psychedelic research encountering strict legal restrictions and negative stigma, the field of psychedelic medicine is now poised for considerable growth in 2023.
As regulations begin to ease, political advocacy increases and research advances, the use of psychedelics in medicine is on its way to becoming a cornerstone in the treatment of mental health and other neurological conditions.
At a psychological level, the use of psychedelic medicines promotes the rewiring of neural circuitry in the brain that helps to combat mental health disorders through behavioral changes and systematic regulation.
Innovative companies have been capitalizing on the advancement of psychedelic substances and their powerful benefits. Private companies have raised hundreds of millions of dollars to develop novel drugs and contribute to research in the field, with many opting to go public for additional funding and support.
With interest in psychedelic medicines on the rise, there is no shortage of opportunities for investors. However, there are many factors to consider before allocating capital to the industry.
The momentum for psychedelic medicines is growing.
Policy changes around medical accessibility and compassionate access.
Exponential leaps forward in scientific discoveries.
Entrepreneurs being empowered to solve big problems.
The psychedelic movement is happening right now.
— Psychedelic Finance (@Psych_Fin) November 13, 2022
The Current Investment Landscape
Recent advancements in psychedelic research and increased support from regulatory entities have prompted a number of psychedelic-based companies to pursue viable solutions to mental health conditions.
Companies such as GH Research and ATAI Life Sciences have gone public with market caps currently over $500 million. Private companies such as Delix Therapeutics and Beckley PsyTech have raised capital totaling over $100 million.
Many of these companies function similarly to biotechnology (BioTech) companies that depend heavily on large investments to fund success in research, clinical trials, and development. As these companies continue to reach Food and Drug Administration (FDA) approval and find product-market fit, the psychedelic medicine market is expected to grow substantially.
Research published by Research and Markets forecasts the Psychedelic Drugs Market to increase from $4.75 billion USD in 2020 to 10.75 billion USD by 2027, representing a compounded annual growth rate (CAGR) of 12.36% during 2021-2027 [1].
 Psychedelic Drug Market Forecast (Source: https://www.visualcapitalist.com/sp/a-visual-guide-to-investing-in-psychedelics/)
Psychedelic Drug Market Forecast (Source: https://www.visualcapitalist.com/sp/a-visual-guide-to-investing-in-psychedelics/) There are a number of sub-sectors that make up the total market, each contributing to the success of psychedelic medicine in different manners. These sectors include:
- Supply and Manufacturing
- Guided Therapy
- Recreational Use
- Delivery Methods
- Consumer Goods
- Research and Clinical Trials
- IP & Patents
The Regulatory Environment
Psychedelics have long been labeled with a negative stigma, and restrictive regulations have reflected those views. However, advancements in medical technology have opened up opportunities for new possibilities.
Advancements in brain imaging technology have helped pave the way for psychedelic research. As scientists more thoroughly understand the impact of psychedelics on the brain, the medicinal benefits of compounds are more easily discovered.
In 2016, we worked with Imperial College to publish results showing, for the 1st time, #psilocybin-assisted therapy could effectively treat treatment-resistant #depression. This has led to FDA Breakthrough Therapy recognition and a Phase II clinical trialhttps://t.co/9CyNMQF2cN
— Beckley Foundation | Psychedelic Research (@BeckleyResearch) November 24, 2022
As the power of psychedelic medicines are more widely understood, regulators have opened up to the possibility of psychedelics being a viable solution in healthcare. In July 2022, the Biden Administration released a statement saying they expect FDA approval for psychedelic-assisted therapies within the next two years [2].
The US National Institute of Health now funds multiple research teams dedicated to psychedelic analogues, or compounds with the benefits of traditional psychedelics but without the side effects [3].
With increased funding and support from regulators, research in psychedelic medicine is experiencing a renaissance not seen since the 1970s.
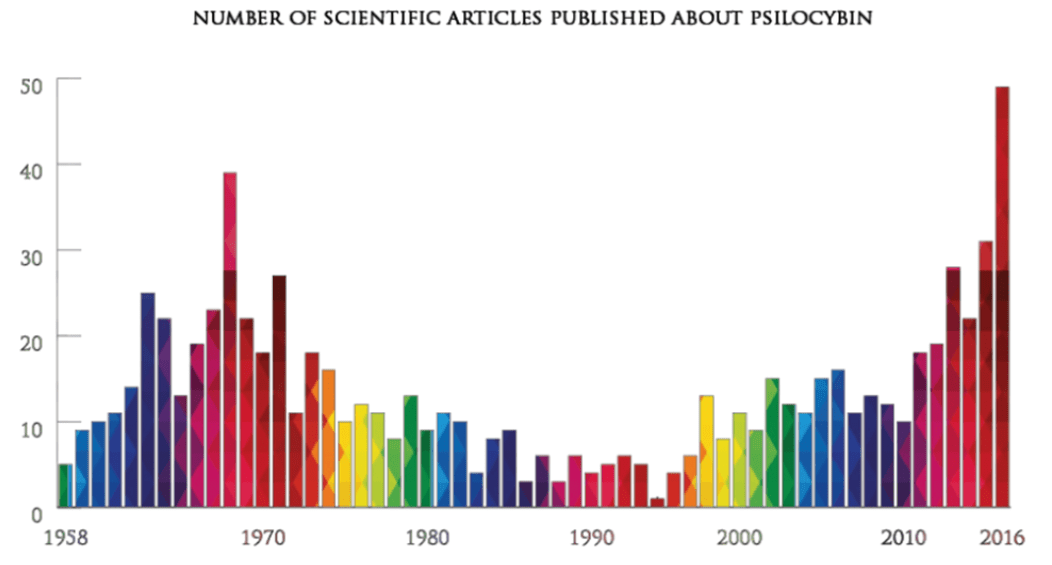 Number of Scientific Articles Published About Psilocybin (Source: https://www.beckleyfoundation.org/psychedelic-research-timeline-2/)
Number of Scientific Articles Published About Psilocybin (Source: https://www.beckleyfoundation.org/psychedelic-research-timeline-2/) As the potential of psychedelics in medicine is uncovered, the FDA has promoted further research by granting “Breakthrough Therapy” status to multiple psychedelic drugs [4]. This designation accelerates the path to drug approval in clinical trials, making the drug development process safer and more efficient.
Although regulations and guidelines around psychedelic research are easing, there are hurdles still to overcome before these treatments are widely available for consumer use.
For example, many of the compounds used are still classified as Schedule 1 drugs by the Drug Enforcement Administration (DEA), meaning they cannot be prescribed by doctors. However, advances in research and FDA approval may prompt reclassification of psychedelic substances.
The Drug Development Process
The pace of innovation and support from important institutions are exciting, but getting these treatments to the end consumer will take time and a lot of money.
All psychedelic drugs must go through the same process to be approved for patient use.
- Clinical Trials
The development of drugs starts with testing through three clinical trials. Each trial consists of increasing guidelines and difficult-to-achieve factors of success. These trials can each take up to 20 weeks before moving to the next stage. The cost of clinical trials for developing drugs can be anywhere from $2 million to $350 million [5].
- FDA Approval
After successful clinical trials, companies may submit their drug for FDA approval. This is a major step in the process, as it largely defines whether the drug will be successful or fail to reach the next stage. Even with successful clinical trials, the FDA may still opt to deny approval due to their stringent guidelines.
- DEA Rescheduling
One of the main reasons why psychedelics find it difficult to reach success in healthcare is because most compounds are classified as Schedule 1 drugs by the DEA. This means that doctors cannot lawfully prescribe these treatments for any purpose. FDA approval for psychedelics may prompt the reclassification of these substances, but the process can be very slow.
It can take years for drugs to reach adoption due to the many tedious steps in the development process. The costs of clinical trials can make up only a fraction of the total cost to bring these drugs to market, with the average cost being around $985 million USD [6].
These hurdles may seem daunting, but an increase in funding and legislative support has shown that they can be achieved. With many drugs already reaching successful clinical trials and FDA approval, there is a path toward the DEA rescheduling of psychedelics for use in medicine.
Although many treatments are still in development, a handful psychedelic-based medicine are currently in progress, including:
- Ketamine-assisted therapy in all states
- Psilocybin-assisted therapy will be available in Oregon starting 2023
- MDMA-assisted therapy may be available by 2023 pending FDA approval
- Advancements in the wide-spread use of the aforementioned substances will pave the way for the approval of compounds such as LSD, ibogaine, and DMT.
Most Popular Compounds Used in Psychedelic Medicine
The span of psychedelics used in medicine has reached beyond just cannabis and psilocybin. With advancements in research of other psychoactive compounds such as DMT, LSD, MDMA, and synthesized psychedelic analogues, the list of psychedelic substances with medicinal properties continues to grow.
Psilocybin
Psilocybin is a psychoactive compound found most commonly in “magic” mushrooms. It produces a strong hallucinogenic effect when ingested but also proves to be an effective treatment for mental health conditions such as depression and addiction.
Psilocybin has recently experienced a friendlier legislative stance which has opened up doors for its use in medicine. Psychedelic startups have made headway by capitalizing on the medicinal benefits while negating the hallucinogenic effect. This increases the accessibility of its medicinal potential to the larger population.
Most commonly administered in micro dosage medicine or in combination with psychotherapy, psilocybin has proven potential to also treat physical illnesses such as lung inflammation, liver disease, and hypothalamic obesity.
In direct clinical trials comparing psilocybin with other antidepressants, Psilocybin has proven to be as effective as the leading Selective Serotonin Reuptake Inhibitor while displaying lower rates of relapse after treatment [7].
Psilocybin is being clinically researched for it’s therapeutic potential around the world.
There has been early clinical validation for many different treatments, including:
– Smoking cessation
– Binge drinking
– Opioid abuse
– Depression
– Cluster headaches
– Anorexia
— Psychedelic Finance (@Psych_Fin) November 26, 2022
Ketamine
Ketamine has been used professionally in healthcare for decades as an anesthetic drug. It is FDA-approved for use as an anesthetic and a nasal spray derivative of ketamine, esketamine, is FDA-approved for treatment-resistant depression (TRD), and major depressive disorder with suicidality..
Ketamine has proven to be an effective anesthetic because of its ability to cause dissociation in patients or the separation of mind from body. As research on the compound progresses, clinicians are finding additional use cases for the treatment of medical conditions such as Parkinson’s disease, mental health disorders, and substance abuse disorders.
Psychedelic startups are taking advantage of recent findings and are developing novel products to treat a wider range of patients outside of a professional setting. Ketamine has also been widely used in combination with other psychedelics, as its effects have proven synergistic when used with other drugs.
Dimethyltryptamine (DMT)
DMT has become a popular compound among psychedelic startups due to its strong effect on serotonergic receptors in the brain. DMT treatments typically work towards disrupting the way the brain processes information and essentially “resetting” patient behaviors. These methods have proven clinically effective for mental health disorders such as depression and anxiety.
Another popular use of DMT is the synthesis of its active compounds, particularly with 5-MeO-DMT. This synthesized version of DMT provides a safer health profile along with quicker treatment time when compared to other psychedelics.
Lysergic Acid Diethylamide (LSD)
LSD was initially synthesized and used in medical settings during the early 1950s to 1970, but harsh regulation hindered its advancement as a viable treatment. Recently, LSD has experienced a softening of legislative boundaries leading to a rise in the research being done to uncover its practical applications within healthcare.
LSD has been clinically proven to provide effective treatment for health conditions such as mental health disorders, pain management, and dementia. Many psychedelic startups seek to minimize the unwanted side effects and isolate its medicinal benefits to make the compound more available to patients.
Other Compounds
Most psychedelic startups have tended to focus on the aforementioned psychedelic compounds, but there are other companies that have found use cases in other substances.
Compounds such as ibogaine, mescaline, and amanita muscaria have all shown promise across a wide swath of health conditions. Some startups, such as Clearmind Medicine, have developed a proprietary compound that combines the benefits of multiple psychedelics.
As research into psychedelic medicine progresses, companies will continue to discover uses for psychedelic medicine outside of the popular compounds, even developing entirely new substances.
Risks of Investing
Although there are numerous opportunities for investment and the potential of psychedelic medicine is exciting, there are some risks to consider before investing.
Regulatory Risk: One of the primary risks facing the psychedelic industry is the compliance of regulators such as the FDA and DEA. Without full approval from the FDA and meaningful reclassification from the DEA, psychedelic-based companies will have difficulties getting their product to market.
Long-Time Horizon: Research in the space is advancing quickly, and investments are flowing into the space even quicker, but psychedelics are still very early in their lifecycle. With long timelines for clinical trials, approvals and reclassification, a world where psychedelic medicine is commonly used by the larger population is still some years ahead.
Competition: As the industry continues to progress, there will continue to be more companies that enter the scene. Although the industry as a whole may succeed, the majority of the competitors will fail. This is why in-depth analysis is so important before investing.
Profitability: Companies can only offer a high return on investment (ROI) to investors if they can eventually make it profitable. With high initial costs for drug development, intense competition, and uncertainties surrounding the psychedelic market, some companies may face a difficult path to profitability.
Keeping up on Twitter
The psychedelic medicine industry moves fast, and it has never been as important (or as easy) for investors to keep track of advancements and new research. Here are 10 of the top Twitter accounts that provide insight and regular updates into the industry.
Palo Santo Fund (@PaloSantoFund): Psychedelic-focused VC firm. Tweets insights from their research and shares insightful information from other industry-leading minds.
Ben Sessa (@BenSessa): Psychedelic researcher and Chief Medical Officer at Awaken Life Sciences. Tweets findings from his research as well as other insightful information circulating the industry.
Psychedelic Finance (@Psych_Fin): A psychedelic finance news outlet helping investors understand and navigate the psychedelics industry. Psychedelic Finance regularly tweets facts, regulatory updates, and insights into industry-leading research.
Michael Haichin (@MichaelHaichin): Founder of the Psychedelic Pharmacists Association and lead editor of the Psychedelic Science Review. Michael tweets regularly, providing thoughtful insight into popular discussions and research pertaining to psychedelic medicine.
Psychedelic Science Review (@psyscireview): A reputable psychedelic and science research institution. They post useful articles published in their journal detailing many of the issues and advancements witnessed in the field.
Johns Hopkins Psychedelic Research Center (@JHPsychedelics): The Center for Psychedelic and Consciousness Research. Regularly posts findings from their studies into how psychedelics affect behavior, cognition, brain function, and other biological markers for health.
David Nutt (@ProfDavidNutt): Chair of Drug Scientist and author of “Drugs – Without Hot Air”, Professor Nutt tweets findings from his own research, all while initiating thoughtful discussions with other professionals on Twitter.
Beckley Foundation (@BeckleyResearch): An industry-leading science institution initiating and funding research into the power of psychedelics in medicine. Beckley posts their own research as well as highlights of other reputable scientific reports.
Drug Science (@Drug_Science): The United Kingdom’s leading independent scientific body on drugs provides reputable research and objective insight into psychedelics and other drugs.
Matthew W. Johnson (@Drug_Researcher): A professor at Johns Hopkins and psychedelic medicine thought leader. Professor Johnson regularly initiates insightful conversations around psychedelic medicine in addition to publishing his own research.
The Potential
The potential of psychedelic medicines for the treatment of mental health and other neurological disorders is advancing quickly. As innovation continues, new companies launch and investments flow in, there will be no shortage of opportunities to capitalize on its success.
There are still risks to consider, especially competition and path to profitability for many companies, but the speed of innovation and the easing of regulations are significant factors in the success of psychedelic medicine.
Understanding the basics of psychedelic medicine, the development process, and the legal/investment landscape are critical to making informed decisions when investing. Always do your own research. Learn as much as you can about a company’s situation and plan for profitability before making a decision.
There will be winners and there will be losers, the amount of effort you put into analyzing companies will decide which side you will be on.

